The Use of 3D-Printed Tissue to Further Brain Tissue Implantation Treatments
Written by Adhvaith Balakrishnan
Edited by Kriti Iyer
April 5, 2024
Edited by Kriti Iyer
April 5, 2024
Research
Imagine you feel a strong headache. You get your brain scanned and find out that you have damaged brain tissue leading to a deadly disease. Would you rather undergo life-threatening brain surgery or possibly have that tissue replaced by a 3D-printed tissue in a faster, safer process?
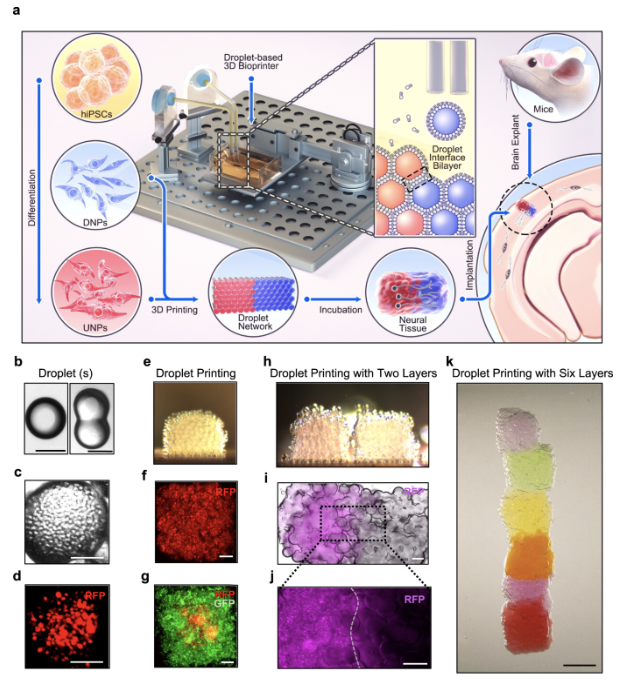
In the past, the study of human tissue through engineering techniques has been difficult, especially to study the cerebral cortex. To combat brain injuries, scientists believed that human induced pluripotent stem cells (hiPSCs) could be utilized. When human tissue dies off, these stem cells have the power to generate a cell type that could fulfill this human tissue (Jin, Y et al., 2023). Though these cells were used in recent advances in stem cell differentiation, their lack of structural control forced scientists to instead hypothesize the idea of implanting tissues that resembled the cellular structure of the damaged brain tissue (Jin, Y et al., 2023).
Through a droplet printing technique, scientists utilized two layers of neural progenitors (NPs), which originated from hiPSCs, and printed them into cerebral cortical tissues. This approach has the potential to advance brain tissue integration and aid in injury or disease treatment (Espuny-Camacho, I et al., 2018 & Mansour, A et al., 2018).
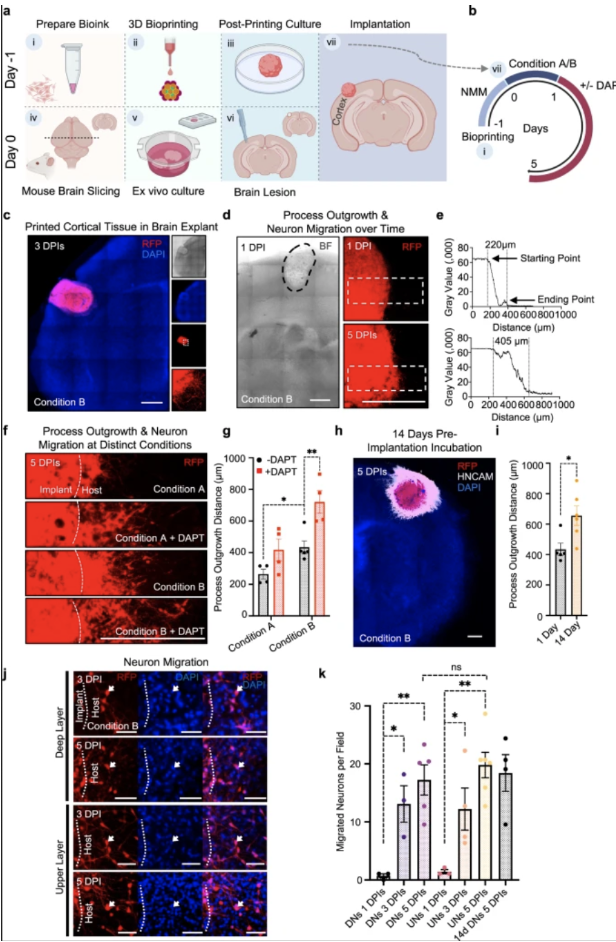
Due to its compatibility for printing and function, progenitors were chosen over mature neurons. Data collected confirmed that the printed two-layer tissues preserved the initial designed cellular structure with stronger capability for cells to grow and migrate (Jin, Y et al., 2023). Once these cortical tissues were printed, they were surgically implanted into different regions of an extracted mouse’s brain to repair damaged tissue. Through fluorescence confocal imaging, the scientists recognized the two indications of printed tissue integration into the brain: neuron migration from the implant towards the rest of the mouse’s brain and process outgrowth (Jin, Y et al., 2023). The third factor was calcium ion activity. The neuron’s display of these “Ca2+ oscillations” establish that there would be communication between the implant and the host (i.e. mouse) (Heck et al., 2007; Grienberger & Konnerth, 2012; Dupont et al., 2006).
Throughout the process, the structure of the two layer tissues were sustained before printing, after printing, and after implantation. Analyzing the process outgrowth, neuron migration, and tissue maturation were key roles in determining if 3D-printed tissue could have a positive, functional effect on brain tissue of living organisms. The observation of Ca2+ oscillations demonstrates a relationship formed between the host (organism) and the printed tissues. Future research could utilize advanced technology, such as progenitors from a human’s own cells, to produce more authentic cortical tissues and treat personal brain damage that is hard to reach (Jin, Y et al., 2023). A limitation was Matrigel being used for the bioink in the tissue printing process as this has certain challenges. However, further studies have reported using Hydrogel as an alternative (Kim et al., 2022). This study is proving to be a breakthrough in using bioengineering tools to treat and diagnose brain tissue problems associated with a disease.
References
Jin, Y et al. (2023, October 4). Integration of 3D-printed cerebral cortical tissue into an ex vivo
lesioned brain slice. Nature News. https://www.nature.com/articles/s41467-023-41356-w
Dupont, E et al. (2005, December 4). Rapid developmental switch in the mechanisms driving
early cortical columnar networks. Nature News. 439, 79-83.
https://www.nature.com/articles/nature04264
Grienberger, C & Konnerth, A (2012, March 8) Imaging calcium in neurons. Neuron. 73(5),
862-885. https://doi.org/10.1016/j.neuron.2012.02.011
Heck H, D et al. (2007, March 5) On-beam synchrony in the cerebellum as the mechanism for the
timing and coordination of movement. PNAS. 104(18).
https://doi.org/10.1073/pnas.0609966104
Kim, S et al. (2022, March 30). Tissue extracellular matrix hydrogels as alternatives to Matrigel
for culturing gastrointestinal organoids. Nature News.
https://www.nature.com/articles/s41467-022-29279-4
Images obtained from articles cited above.